Tech
Researchers develop photocatalyst with irregular surface characteristics to convert carbon dioxide into fuel
× close
A research team have developed a high-efficiency photocatalyst that converts carbon dioxide (CO2), a major contributor to climate change, into natural gas (methane, CH4). The team is led by Professor In Soo-il of the Department of Energy Science & Engineering at DGIST. The study is published in the journal Applied Catalysis B: Environment and Energy.
Global warming causes abnormal climates around the world, threatening the survival of the human race. Reducing greenhouse gases is crucial to solving the increasingly concerning problem of global warming, which requires converting atmospheric carbon dioxide into other substances.
Photocatalytic technology is an environmentally friendly solution that converts carbon dioxide into useful substances such as natural gas using only solar energy and water. The natural gas produced can be used in our daily lives as a fuel for heating and cooling systems and vehicles.
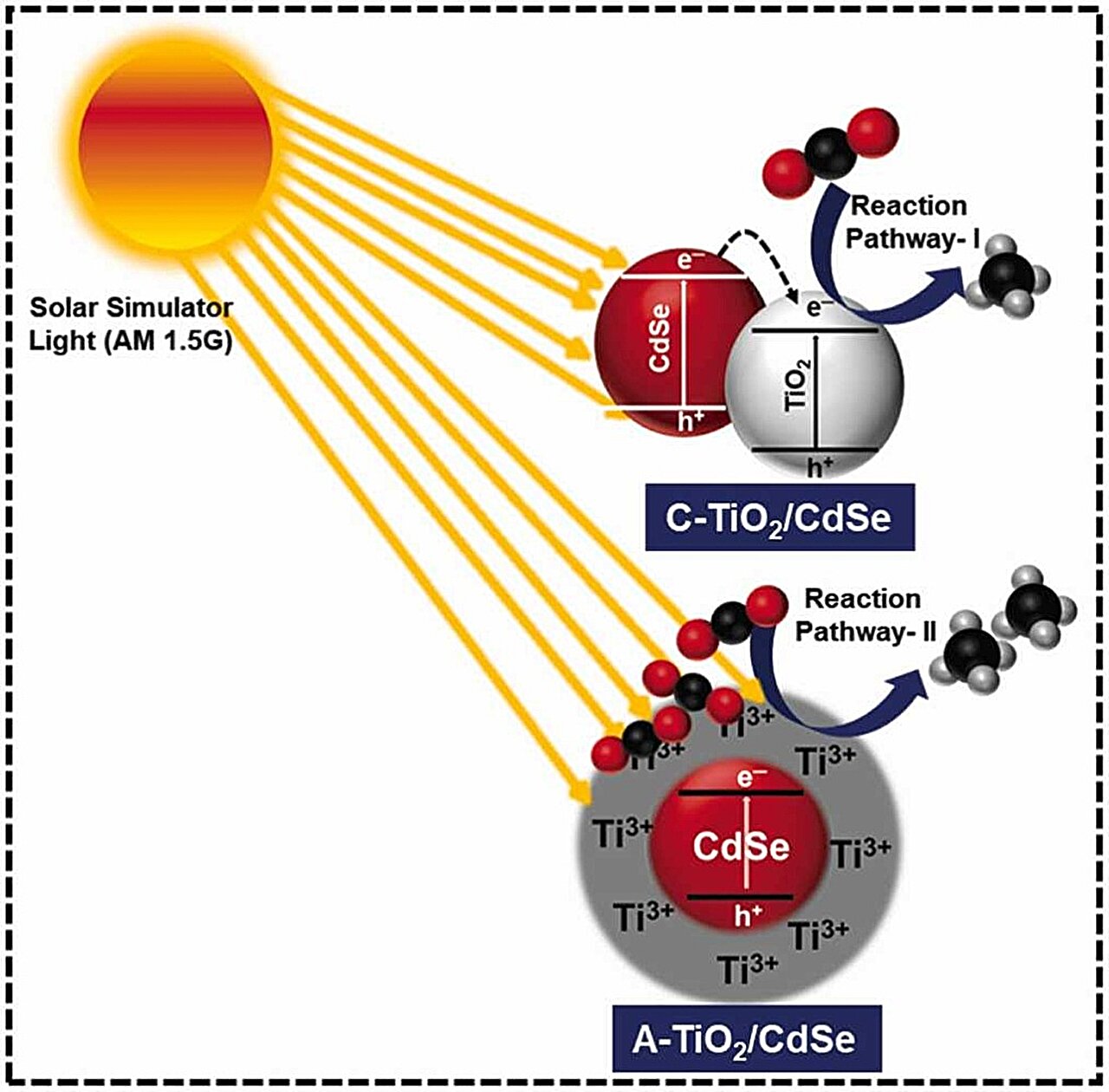
The research team combined cadmium selenide, which absorbs visible and infrared light, with titanium dioxide—a metal oxide and well-known photocatalytic material—to convert carbon dioxide into natural gas with high efficiency.
Previously, crystalline titanium dioxide, which has a periodic lattice structure, was analyzed as a photocatalytic material. However, the formation of active sites for the trivalent cations of titanium (Ti3+) was limited owing to the regular arrangement of the particles.
To overcome this problem, Professor In’s team improved the catalytic reaction using amorphous titanium dioxide, which can form more active sites for Ti3+ through irregular particle arrangements that lack the periodicity of the lattice structure.
In addition to improved catalysis, the charge-transfer process is stable, ensuring a sufficient supply of electrons to participate in the reaction. This facilitates the conversion of carbon dioxide into carbon compounds, particularly methane fuel. Furthermore, unlike conventional photocatalysts that require high temperatures for regeneration, amorphous catalysts can be regenerated within one minute when oxygen is supplied to the reactor without heating.
The research team’s newly developed amorphous titanium dioxide–cadmium selenide photocatalyst (TiO2-CdSe) maintained a methane-conversion performance of 99.3% for the first 6 hours after 18 hours of photoreaction, making it 4.22 times more regenerative than the crystalline photocatalyst (C-TiO2-CdSe) having the same composition.
“This study is significant in that we have developed a catalyst with regenerative active sites and identified the mechanism by which carbon dioxide is converted into methane using an amorphous catalyst through computational chemistry research,” said DGIST Professor In.
“We will conduct follow-up research to improve the energy loss of the amorphous photocatalyst and enhance its long-term stability for future commercialization of the technology,” he added.
More information:
Niket S. Powar et al, Unravelling the effect of Ti3+/Ti4+ active sites dynamic on reaction pathways in direct gas-solid-phase CO2 photoreduction, Applied Catalysis B: Environment and Energy (2024). DOI: 10.1016/j.apcatb.2024.124006
Provided by
DGIST (Daegu Gyeongbuk Institute of Science and Technology)










