Fitness
Researchers discover novel method to predict effectiveness of mesenchymal stromal cells for cartilage repair
× close
Researchers have discovered a more efficient method for evaluating the ability of mesenchymal stromal cells (MSCs) to regenerate cartilage. Their novel method is also the first to utilize topological defects in the self-assembly of MSCs to forecast their cartilage regeneration potential.
MSCs are promising therapeutic agents for cartilage regeneration, and they have the potential to help create new cartilage tissue within the body. Previously, assessing the potential of these cells for effective cartilage regeneration involved culturing them in a complex 3D environment for a lengthy period of 21 days.
The novel method, developed by researchers from Critical Analytics for Manufacturing Personalized-medicine (CAMP) Interdisciplinary Research Group (IRG) at Singapore-MIT Alliance for Research and Technology (SMART), and collaborators, involves a simpler 2D monolayer culture system of the cells for nine days, followed by imaging and processing of the images. The method is non-destructive, more accurate, and faster—requiring only nine days. This could significantly accelerate the development of cartilage regeneration therapies.
“This innovative approach to evaluating MSC effectiveness for cartilage repair represents a significant advancement by enabling non-destructive evaluation of MSCs and reducing the assessment period to a mere nine days,” stated Dr. Ekta Makhija, lead author of the paper and Research Scientist at SMART CAMP at the time of the study.
× close
In the paper “Topological defects in self-assembled patterns of mesenchymal stromal cells in vitro are predictive attributes of condensation and chondrogenesis,” published in the journal PLOS ONE, SMART CAMP demonstrated that topological defects in MSC self-assembly could indicate their potential for regenerating cartilage.
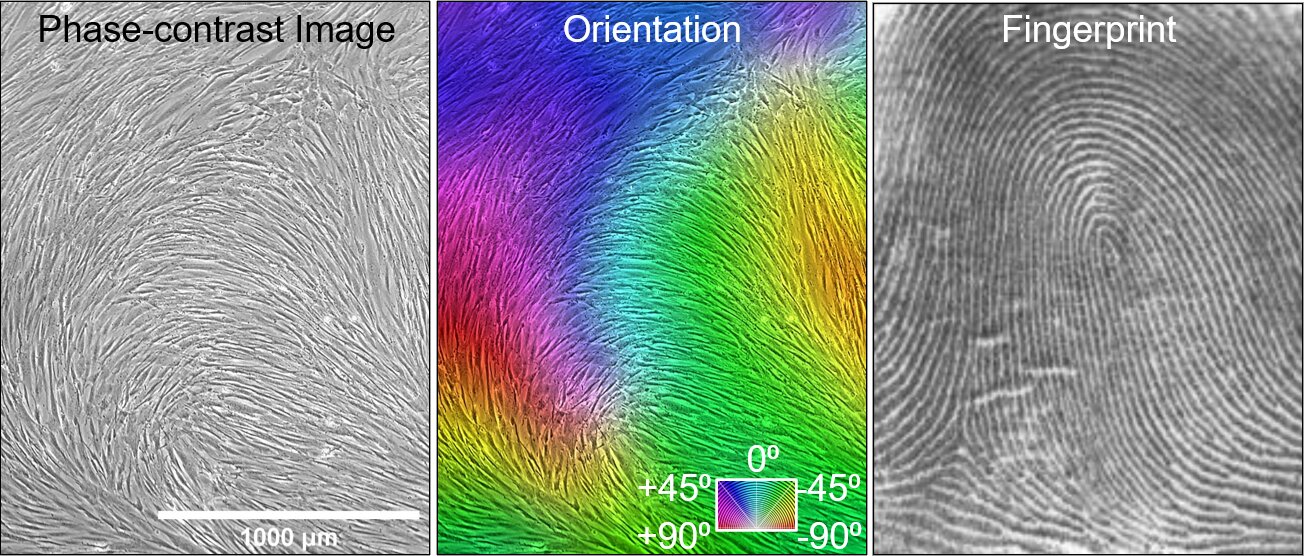
The researchers captured images of the cells on days three, six, and nine, then analyzed the patterns formed by the cells as they self-assembled. By day nine, the researchers noted that certain aspects of these patterns resembled liquid crystal structures with identifiable topological defects, such as irregularities or imperfections in the patterns.
These defects in the self-assembled patterns matched fingerprint-like patterns that align with cartilage markers, known as mesenchymal condensations, observed during early cartilage development stages. These patterns serve as indications of the cells’ potential for cartilage formation and act as reliable markers for cartilage regeneration.
This innovative approach shows promise for greater accuracy and reliability compared to traditional methods, which involve a 21-day process of growing MSCs in a specialized 3D setting and then assessing specific proteins indicating cartilage growth.
Similar to an initial evaluation determining whether a particular batch of MSCs possesses the requisite “tools” and cooperative abilities needed to repair damaged cartilage effectively, the new research identified factors predicting MSCs’ ability to form and regenerate cartilage, building upon prior research in 2014 under the BioSystems and Micromechanics (BioSyM) IRG at SMART.
Ten years on and with significant advancements in the field, SMART researchers are now focusing on the collective behavior of MSCs when grouped together rather than examining cells in isolation. These collective patterns serve as a distinctive signature for the cell population, offering a potentially more dependable and more accurate method for predicting how effectively the cells collaborate in regenerating cartilage.
This faster, non-destructive and simpler approach allows for the assessment of a larger number of MSCs, potentially pinpointing the most efficacious ones for future cartilage regeneration therapies.
“This innovative method holds a major advantage as it allows manufacturers to conduct more frequent testing of their cell-based medicine, ensuring safety, purity, and effectiveness throughout production. This can potentially speed up the traditionally long process of securing regulatory approval for cell-based medicines,” added Dr. Zhiyong Poon, Principal Investigator at SMART CAMP, Senior Research Fellow at Singapore General Hospital (SGH) and a corresponding author of the paper.
“Our new method marks a substantial advancement in the way we approach cartilage regeneration. By offering a more efficient means of evaluating MSCs in production, we can accelerate the development of therapies for joint injuries and common ailments, particularly those affecting the aging population, such as osteoarthritis, and address limitations of current surgical and pharmaceutical approaches in restoring cartilage function,” added Professor Eng Hin Lee, Principal Investigator at SMART CAMP, Emeritus Professor at the National University of Singapore (NUS) Yong Loo Lin School of Medicine and one of the authors of the paper.
Having established a successful and novel method of predicting the effectiveness of MSCs for cartilage repair in vitro through the observation of topological patterns, SMART researchers’ next steps will involve assessing whether these patterns are similarly predictive of MSC effectiveness for cartilage repair in vivo.
More information:
Ekta Makhija et al, Topological defects in self-assembled patterns of mesenchymal stromal cells in vitro are predictive attributes of condensation and chondrogenesis, PLOS ONE (2024). DOI: 10.1371/journal.pone.0297769
Journal information:
PLoS ONE










